Discovery and structure-guided fragment-linking of 4-(2,3-dichlorobenzoyl)-1-methyl-pyrrole-2-carboxamide as a pyruvate kinase M2 activator
Matsui, Y., Yasumatsu, I., Asahi, T., Kitamura, T., Kanai, K., Ubukata, O., Hayasaka, H., Takaishi, S., Hanzawa, H., Katakura, S.(2017) Bioorg Med Chem 25: 3540-3546
- PubMed: 28511909
- DOI: https://doi.org/10.1016/j.bmc.2017.05.004
- Primary Citation of Related Structures:
5X1V, 5X1W - PubMed Abstract:
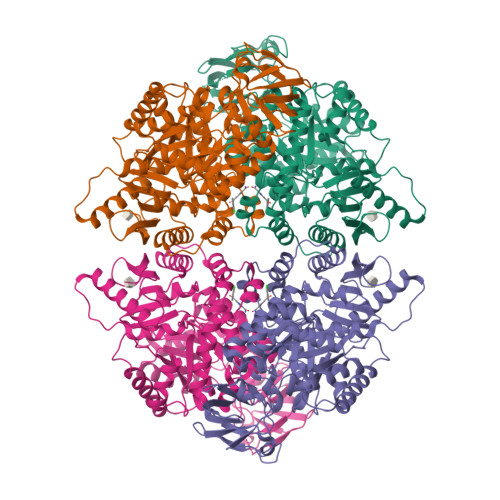
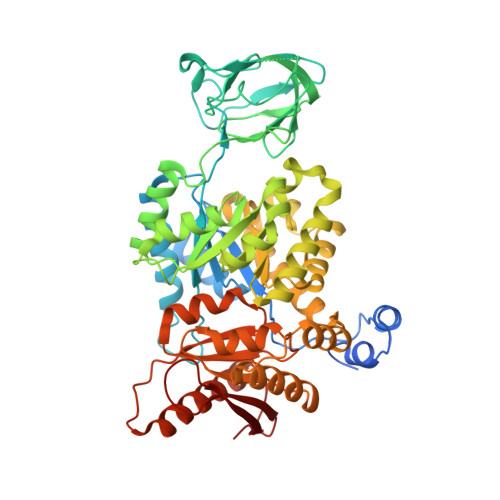
Tumor cells switch glucose metabolism to aerobic glycolysis by expressing the pyruvate kinase M2 isoform (PKM2) in a low active form, providing glycolytic intermediates as building blocks for biosynthetic processes, and thereby supporting cell proliferation. Activation of PKM2 should invert aerobic glycolysis to an oxidative metabolism and prevent cancer growth. Thus, PKM2 has gained attention as a promising cancer therapy target. To obtain novel PKM2 activators, we conducted a high-throughput screening (HTS). Among several hit compounds, a fragment-like hit compound with low potency but high ligand efficiency was identified. Two molecules of the hit compound bound at one activator binding site, and the molecules were linked based on the crystal structure. Since this linkage succeeded in maintaining the original position of the hit compound, the obtained compound exhibited highly improved potency in an in vitro assay. The linked compound also showed PKM2 activating activity in a cell based assay, and cellular growth inhibition of the A549 cancer cell line. Discovery of this novel scaffold and binding mode of the linked compound provides a valuable platform for the structure-guided design of PKM2 activators.
Organizational Affiliation:
Daiichi Sankyo RD Novare Co., Ltd., 1-16-13, Kita-Kasai, Edogawa-ku, Tokyo 134-8630, Japan. Electronic address: matsui.yumi.gk@rdn.daiichisankyo.co.jp.